"Termite guts:
the world's smallest bioreactors"
the world's smallest bioreactors"
Brune 1998
Lignocellulose and Lignocellulose Digestion
|
|
Lignocellulose
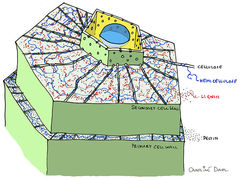
While the contents of a plant cell are fully digestible, the cell wall that encloses them provides strength and support and, as such, is primarily composed of strongly bound, interconnected components such as cellulose, hemicellulose and lignin (Image 5). Cellulose is composed of repeating units of cellobiose. This bond cannot be broken by mammalian enzymes but can be broken by insect or microbial enzymes (McDonald et al. 2011). Hemicellulose is composed of different sugar units using a variety of bonds. Lignin is not a carbohydrate but has a complex structure that can make very strong bonds with other plant materials dramatically increasing their resistance to digestion.
|
|
Every year, plants produce approximately 2 – 5 x 1012 metric tons of lignocellulose around the world (Glazer and Nikaido 2007 cited in Sun and Zhou 2011). Wood is approximately 20% lignin, 25% hemicellulose and 40% cellulose (Scharf et al. 2011a). While forage plants are more digestible, lignocellulose bonding generally increases as plants age.
Hundreds of fungi and bacteria and at least a hundred insect species can digest lignocellulose to varying degrees (Table 1) (Sun and Zhou 2011). Ruminants (cows, goats, etc.) are better than monogastrics (horses etc.) at digesting cellulose and hemicellulose with the help of their intestinal microorganisms and fermentation ability but are relatively poor utilisers compared to insects. |
Table 1: Insect cellulose digestion
|
Lignocellulose Digestion
|
Due to its recalcitrant properties, lignocellulose digestion is a combination of the following factors (Breznak and Brune 1994):-
a. physical - chewing and grinding into smaller pieces by mandibles (Image 6) and gizzard to increase surface area for attack; b. chemical – the presence of termite enzymes (particularly cellulases), microaerobic or anoxic conditions and highly alkalinic (pH>12) conditions; and, c. biological processes - bacteria, archaea and protozoa (in lower termites) that attack food particles with their own enzymes. |
For nearly a century, researchers assumed termite lignocellulose digestion was facilitated by symbionts, such as bacteria, archaea and protozoa but a variety of digestive enzymes (cellulases) have since been discovered throughout the termite digestive system (Scharf et al. 2011b). Termite digestive enzymes contribute significantly to termite metabolism with Scharf et al. (2011b) estimating a third of glucose and 40% of pentose in Reticulitermes flavipes was released via host enzymes while the rest was released via symbiont enzymes. There are three main types of cellulase in termites (Lo et al. 2011):
a. endo-β-1,4-glucanases – hydrolyse (break) internal bonds (found in most living organisms);
b. cellobiohydrolases – hydrolyse cellobiose from ends of cellulose chains (found in protozoa, fungi and bacteria); and,
c. β-glucosidases – convert short sugars to glucose.
In combination, these cellulases can increase glucose release from lignocellulose or hemicellulose, 200-300 times or 70-100 times greater, respectively, than a single cellulase (Scharf et al. 2011b). Generally cellulase levels are higher in wood feeders than in soil or fungus feeders (Lo et al. 2011).
a. endo-β-1,4-glucanases – hydrolyse (break) internal bonds (found in most living organisms);
b. cellobiohydrolases – hydrolyse cellobiose from ends of cellulose chains (found in protozoa, fungi and bacteria); and,
c. β-glucosidases – convert short sugars to glucose.
In combination, these cellulases can increase glucose release from lignocellulose or hemicellulose, 200-300 times or 70-100 times greater, respectively, than a single cellulase (Scharf et al. 2011b). Generally cellulase levels are higher in wood feeders than in soil or fungus feeders (Lo et al. 2011).
|
Wood-feeding termites (Image 7) tend to have a more diverse microbiota than the non-wood feeding termites, possibly due to the greater demands of lignocellulose digestion (Boucias etal. 2013). R. flavipes has thousands of prokaryotic symbionts (99.9% are bacteria and 0.11% are archaea) (Boucias et al. 2013) and 12 to 13 species of protozoa (Bruggerolle and Radek (2006) cited in Scharf et al. 2011b). Its microbiota also show good resiliency, with only a 12% variation in microbial species even after 7 days on an alternative lignocellulose diet (Boucias et al. 2013).
|
|
The fungus feeders (Macrotermitinae) (Image 8) have a fairly simple digestive structure probably because of their mutualistic relationship with fungi. They grow fungus on their lignin-rich faeces. Then, after the fungus has digested the lignin, the termite eats the fungus - thus simplifying the whole digestive process.
|
 Image 9. Anaerobic biodigester, Tel Aviv, Israel. Image from Wikimedia.
Image 9. Anaerobic biodigester, Tel Aviv, Israel. Image from Wikimedia.
Lignocellulose as a Potential Energy source
Lignocellulose is also being investigated as an energy source. First generation biofuels are made from food materials whereas second generation biofuels are intended to exclude non-food crops and include crop residues and other plant biomass, in particular some of the massive amounts of lignocellulose growing each year (Sun and Zhou 2011). Currently lignocellulose has to undergo gasification, pyrolysis or hydrolysis to produce biofuel (Scharf and Tartar 2008). Due to its complex nature lignocellulose requires significant energy, particularly in the first two processes, whereas the final process, hydrolysis, holds the potential for cheaper, easier production using cellulases. Human-built anaerobic digesters (Image 9) are only 1/3 as efficient as rumen digestion and even less efficient than termite digestion (Bayane and Guiot 2011). Insect and mammalian lignocellulosic digesters provide optimal environmental conditions for microbial fermentation (that can be replicated, at least partially, in human biodigesters) but also include other processes that assist digestion, such as (specifically in termites): oxidation gradient, alkaline environment and transit time (Bayane and Guiot 2011). Termites, and their symbionts are therefore being studied for ways of simplifying and reducing the cost of conversion to biofuel.
Lignocellulose is also being investigated as an energy source. First generation biofuels are made from food materials whereas second generation biofuels are intended to exclude non-food crops and include crop residues and other plant biomass, in particular some of the massive amounts of lignocellulose growing each year (Sun and Zhou 2011). Currently lignocellulose has to undergo gasification, pyrolysis or hydrolysis to produce biofuel (Scharf and Tartar 2008). Due to its complex nature lignocellulose requires significant energy, particularly in the first two processes, whereas the final process, hydrolysis, holds the potential for cheaper, easier production using cellulases. Human-built anaerobic digesters (Image 9) are only 1/3 as efficient as rumen digestion and even less efficient than termite digestion (Bayane and Guiot 2011). Insect and mammalian lignocellulosic digesters provide optimal environmental conditions for microbial fermentation (that can be replicated, at least partially, in human biodigesters) but also include other processes that assist digestion, such as (specifically in termites): oxidation gradient, alkaline environment and transit time (Bayane and Guiot 2011). Termites, and their symbionts are therefore being studied for ways of simplifying and reducing the cost of conversion to biofuel.